Medical Device Software Design and How to Build It: The Complete Guide

In this article, we’ll discuss custom medical device software development. The new generation of healthcare startups and companies offers innovative, portable, convenient medical devices for a variety of healthcare situations and needs. The relatively new concept of “Software as a Medical Device” (SaMD) is unveiling huge opportunities in the creation of advanced applications for existing or prototyped medical devices.
| ⚠️ Please contact our experts for a deeper (free) consultation on medical device software design and development in your health-tech projects or healthcare organization! |
When we’re talking about a typical medical device or piece of healthcare equipment, most people usually imagine something like this…
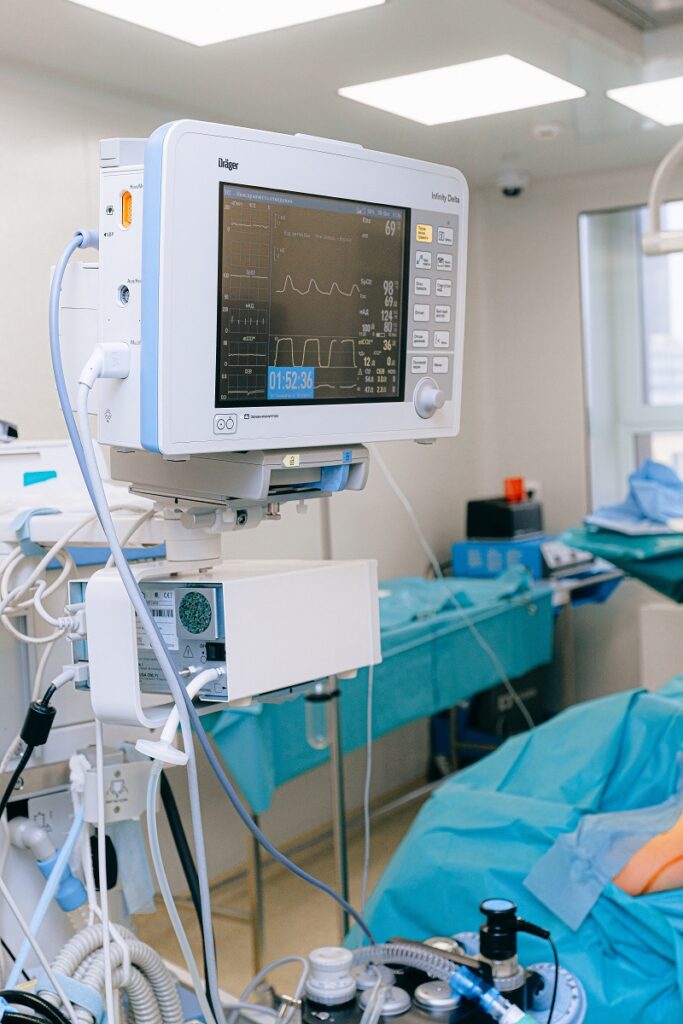
…a piece of cumbersome, complicated, professional-grade medical equipment with mind-boggling settings/controls/wires that’s barely usable anywhere outside hospital walls. Is this concept of healthcare devices still relevant?
For many years, this type of image of medical equipment has been prevalent not only with the general public, but with physicians, too. Anything smaller or less complicated has long been perceived as unreliable, suspicious, and generally not worth the attention of serious healthcare professionals…for a good reason!
Progress, however, does not stand still.
Thanks to ongoing advancements in electronics and IT, more and more models of modern healthcare equipment are being designed and produced smaller, more easily portable, easier to manage, and (finally!) readily available for use in almost any location beyond the clinical setting, including patients’ homes.
What’s more important, even miniature medical devices are now efficient and reliable enough to be employed for vital healthcare practices and processes.
The shocking truth about legacy operating systems driving medical devices…
A group of hi-tech researchers recently revealed that about 83% of medical IoT devices in the United States are driven by obsolete (legacy) or unsupported software systems. Decommissioned, abandoned by vendors, and insecure, these operating systems are highly vulnerable to exploits and cybercrime because they keep nearly 98% of sensitive medical data traffic exposed or inefficiently encrypted. This makes the issue of upgrading or modernization of outdated healthcare applications very acute and urgent. If you require deep re-engineering and rebuilding of your medical equipment operating systems, please contact our team for finding a potential solution! Learn more about us…

| ? Who are we? TATEEDA GLOBAL is a custom medical device software development company that can help you with software design for medical devices. We have vast experience with U.S. medical startups and companies offering portable devices, including remote patient monitoring and custom telemedicine solutions. Learn about our experience with ECG-monitoring biosensor software development ⇒ If you are interested in medical device software developers, feel free to consider us: ✅ We are headquartered in the U.S. (San Diego, CA) and have local project experts and software engineers available for personalized communications and technical consulting. ✅ We offer a convenient time zone for North American clients, dedicated project managers, and favorable project rates, thanks to our well-organized R&D branch in Ukraine, LATAM nations, like Brazil and Columbia, and other nearshore custom software development destinations. ✅ We are open for free tech consulting. Do you need expert advice on technologies for your medical device software development? Please reach out to our medical software engineers today ⇒ |
Table of Contents
Who May Need Software Development for Medical Devices?
Medtech software development can be demanded by many types of companies, ranging from established Medical Device Manufacturers to innovative Healthtech Startups. These companies may seek specialized programming for medical devices, offshore medical device app development, and comprehensive medical device development services to enhance their products and services.
| Medical Device Manufacturers | Companies that specialize in manufacturing medical devices and need software solutions to enhance their products, focusing on programming medical devices and medical device application development. |
| Healthcare Institutions and Hospitals | Organizations seeking custom software for medical devices used within their facilities for patient care, diagnostics, or monitoring. |
| Biotechnology Companies | Firms involved in biotech research and development that require software solutions for their medical devices, emphasizing programming medical devices for enhanced functionality. |
| Telehealth Service Providers | Companies offering telehealth services that need software for medical devices to facilitate remote patient monitoring and consultations, with a focus on medical device app development. |
| Pharmaceutical Companies | Organizations involved in pharmaceuticals requiring software solutions for medical devices related to drug delivery systems or clinical trials, emphasizing comprehensive medical device development services. |
| Healthtech Startups | Emerging companies focused on innovative healthcare solutions, including medical devices, requiring software development for medical devices and medical device app development. |
| Research and Development Labs | Emerging companies focused on innovative healthcare solutions, including medical devices, requiring software development for medical devices and medical device app development. |
| Regulatory Compliance Consultants | Consultants or firms assisting medical device manufacturers in ensuring compliance with industry regulations through software solutions, emphasizing comprehensive medical device development services. |
| Clinical Research Organizations (CROs) | Organizations conducting clinical trials that need software for medical devices used in research, with a focus on programming medical devices for data collection and analysis. |
| Diagnostic Equipment Manufacturers | Companies specializing in diagnostic equipment that require software solutions to enhance the functionality of their devices, emphasizing medical device app development and programming medical devices. |

Are you looking for software developers for medical devices?
If you’re a healthcare company needing services like medical device application development or support in the software development process for medical devices, TATEEDA can assist. Our tailored project outsourcing and staff augmentation services cater to the unique needs of medical device manufacturers, healthtech startups, and telehealth service providers, ensuring efficient solutions to enhance your capabilities.
Different Types of Software for Medical Devices
There are different software classifications specific to medical equipment. Before starting a new project, it’s necessary to clearly define the requirements for your medical device software development, as the scope and technologies can be pretty diverse, including embedded coding and SaMD.
However, the majority of real-life medical equipment startups and projects are looking to employ a mixed set of technologies. Let’s learn more about the major domains of software development for medical devices…
Embedded Medical Systems and Embedded Medical Software Development
This domain encompasses low-level programming of micro-components—like microcontrollers and micro-chips—with microprocessors and embedded memory. This can all be found under the hood of most healthcare equipment. Some examples of medical devices featuring embedded systems driven/configured by embedded code are:
- pulse oximeters
- electronic defibrillators
- smart (bio)sensors
- automated infusion pumps
- glucometers
- electronic thermometers
- electronic blood pressure sensors
- medical imaging equipment: X-ray, ECG, EEG, MRI, CT, etc.
- a great variety of lab equipment.
| What are the programming languages used in medical devices? Embedded medical software is an integrated and indispensable part of healthcare devices, especially in terms of electronics and electromechanical parts. The embedded code should be highly optimized and reliable, utilizing minimum program memory (because of the limited computing capacities of microelectronic components). Languages used in embedded healthcare device programming include C, C++, Python, MicroPython, and Java. |

Do You Need Help with Medical Device Software Design?
With us, you can hire experienced IT engineers to develop software for medical devices and complex digital health systems.
Embedded programming is crucial for healthcare equipment and biomedical applications, as it regulates the usage of various electronic components and helps integrate medical devices with non-specific or general-purpose software and hardware, including PCs, EHRs, Wi-Fi, and many other systems.
While embedded system development for some medical devices does require only primitive programming skills, other projects require advanced competence in healthcare device engineering. Just imagine what it takes to program and calibrate all the embedded electronics in a large, sophisticated machine like a modern MRI tomograph.
TATEEDA GLOBAL’s engineering team has deep experience with embedded systems in healthcare and lab equipment programming. If you need a free estimate or consultation regarding any type of medical device programming including health-tech startups, biotech facilities, or other life-science solutions, please contact us.
Learn more: How to Build a Lab Information Management System (LIMS)
Software as a Medical Device (SaMD)
This realm of medical device software development covers all products and solutions that don’t need a specific medical device. For example, SaMD systems run on non-specific medical platforms, including laptops, desktops (learn why desktop applications always matter), mobile devices, etc.
This type of software usually performs one or several functions related to:
- medical data visualization and representation
- medical data processing and interpretation
- medical data management and storage
- specific configuration and technical diagnostics of certain medical devices
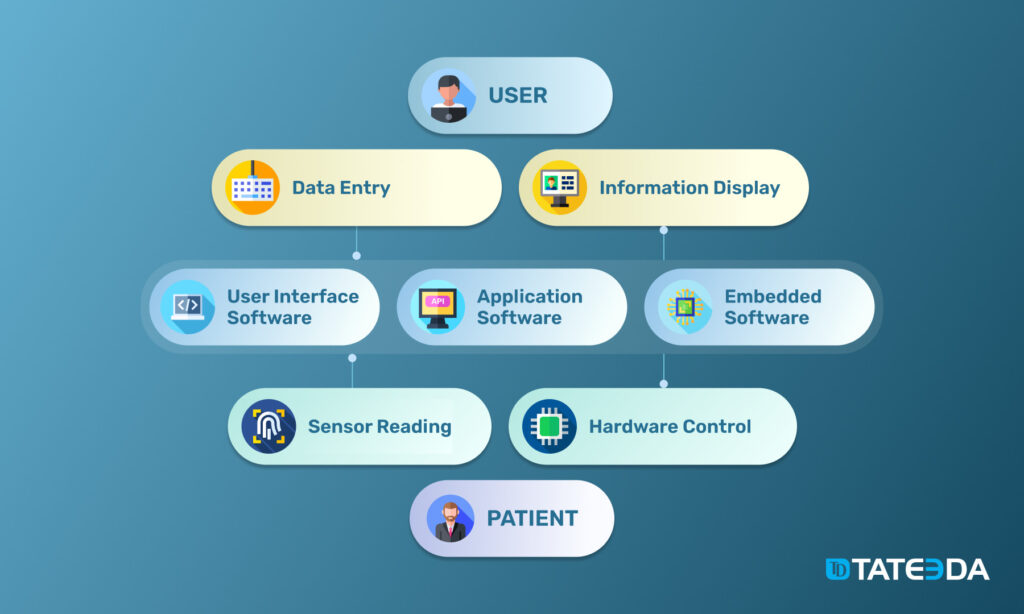
Basically, software as a medical device is characterized by the following features:
- SaMD is not a part of medical equipment, and it is not required for a medical device to execute its functions or tasks.
- SaMD runs on general-purpose computers and infrastructure.
- It can interface with certain dedicated medical devices including devices’ inner/embedded components and embedded coding to receive data obtained by the device or to manage its functionality.
| Software as a medical device can use a variety of tools, including every kind of specific (e.g., purely medical scanners or sensors) or non-specific devices (like web cameras or microphones.) Thanks to general-purpose platforms that run SaMD solutions, a great variety of programming languages and technologies can be used, including web-based and mobile development. All types of SaMD fall within I-IV categories according to the severity of patient conditions they are intended to deal with, as offered by the International Medical Device Regulators Forum. |
Software development for medical devices, including the SaMD concept, requires specific skills and expertise, which are not easily available in the American market. If you are looking for qualified assistance with a medical device programming project, please make sure to contact our seasoned healthcare technology expert to discuss the details and learn more about our capabilities regarding medical device coding and other software solutions. Book a free call today:

Slava Khristich
Healthtech CTO
Based in San Diego, Slava knows how to design an efficient software solution for healthcare, including IoT, Cloud, and embedded systems.
Some examples of Software as a Medical Device (SaMD) include:
- Patient imaging or scan analysis: Software that analyzes patient data to discover patterns, signs, or trends helps physicians to better identify barely noticeable yet important changes in health conditions and/or facilitate diagnosis and treatment. For example, this could include support in decision-making for correct differentiation between ischemic and hemorrhagic stroke in patients with acute stroke conditions. The outcome has the potential to affect choice of therapy and/or medical intervention greatly.
- Sound monitoring for sleep apnea prevention: A smart device microphone can be used to automatically detect interrupted breathing during sleep and trigger an alarm to wake the sleeper. The same technology can be used to automatically detect unusual breathing patterns and send an alarm to emergency responders if dangerous conditions are detected (this can work for single and/or elderly patients.) Learn more: How to Develop a Senior Care Mobile App
- Remote ECG-monitoring: Adhesive or implantable sensors can be used to remotely observe cardiac patients and record their heartbeat patterns while detecting unusual or life-threatening ECG patterns and/or events (arrhythmia, bradycardia, etc.) that should be immediately reported. Learn more: Virtual Nurse App Development Guide
- Medical data-viewing applications: These include any type of software that helps healthcare professionals access, check, visualize, share, record, and/or interpret health data received from specific bioelectronic sensors or medical devices, including multiple metrics like heart rate, blood pressure, skin temperature, and more.
Do you need help with custom medical device software development?
If you’re looking for a medical device software development company that can partner with you to help you carry out an innovative medical equipment project or define your project requirements and technologies, please contact TATEEDA GLOBAL for free consultation.
We have extensive experience in the following areas:
- Programming medical devices and integration of software systems with automated lab and medical equipment based on up-to-date IT interoperability standards in healthcare
- Development of mobile and desktop user interfaces for medical devices and laboratory instrumentation
- Building client-server applications for obtaining, accumulating, and managing medical data collected from medical and lab equipment
- Embedded programming for micro-components
- IoT and Cloud solutions for healthcare systems
- Integration of medical devices with business software solutions, like healthcare ERP.
We offer skilled medical technology developers, favorable project rates, project augmentation services, and convenient communication channels for our customers in the North American time zone. Learn more about our custom software development services in San Diego and services here:
Custom Healthcare Solutions
See how we can engineer healthcare software, validate your ideas, and manage project costs for you.

How to Build Custom Medical Device Software
Of course, medical equipment software development requires considerable engineering effort and knowledge. It starts with defining project goals and scope while creating a profound medical device/software architecture. Let’s discuss the most important aspects of building medical device software…
A High-level View of the Medical Device App Development Process
SaMD development and embedded system development are associated with a wide range of medical device software development processes. That’s why they require different specialists, diverse engineering expertise, and well-qualified health-tech engineers to amalgamate the results.
Basically, from a top-level view, a medical device software engineering project requires the following actions:
- Define the technological stack of your medical device software project: Is it a purely embedded/SaMD project, or mixed?
- Identify and fulfill the FDA requirements for your type of medical device and software (in compliance with HIPAA.) Certain types, especially new medical equipment, require ISO certifications and compliance with other standards.
- Based on your project scope, hire or gather a team of developers capable of embedded and/or conventional software development, which is required for SaMD development.
- Let a health-tech engineer design your software/device system specifications.
- Make sure a skilled project manager is assigned to break down the project into distinct phases and tasks.
- Assign these tasks to the right specialists: health-tech UI/UX designers, back-end/front-end developers, QA specialists, and others.
- Make sure you have medical professionals available for consulting and testing. Remember that medical staff (physicians, nurses, surgeons) and their patients are expected to be the end-users of your medical product, so their interests should be represented.
Tech Stack to Use in Medical Device Software Development
There exists a great abundance of technical solutions and tools that can work for medical equipment software development. Your specific configuration depends on your project and its scope (which should be discussed and specified with your vendor). If you want to discuss specific tech options and the medical device software development process with an expert, please contact us for a free consultation.
The average tech stack used in medical device software engineering can include a mix of anything from the list below:
- Integrated Development Environments (IDE) and Compiler
- Embedded coding languages: C, C++, Python, MicroPython, Java
- Debug Devices and Software (Debugger)
- Emulators
- Testing Software and Devices
- Cloud development: Azure, AWS, Digital Ocean, Google
- Integrated health tech solutions with wearables, mobile, and IoT devices
- Front-end development: Angular, React, Vue, Node.js, and Core JavaScript
- Supportive Software: C#, Node.js, JavaScript, TypeScript, Dart, PHP, and SQL
- Middleware and additional modules from a variety of vendors: 3D imaging, 3D printing, AI, ML, AR/VR, medical SaaS, and more.
| ? How can we help? TATEEDA GLOBAL is a skilled IT consulting and software development vendor that features significant expertise in different tech fields including custom IoT solution engineering, DevOps services, biotech software services, and .NET development & testing. Please see our project portfolio for more details: |
Delivered Healthcare Software Portfolio
The leading American healthcare companies benefit from working with us.
How to Choose the Right Development Partner
Do you need to build your medical device software from scratch? Or do you have an ongoing project that requires additional augmentation and resources? Finding and choosing the right medical device software developers can be a very challenging task. Here are a few recommendations on hiring a good team for your project:
- Before approaching any medical technology developers, make sure you have a sufficient plan for your medical equipment software (or at least a clear vision of your future medical product). Having a ready plan or well-elaborated vision will save you a great deal of time when dealing with software professionals.
- Approach only reputable companies. Check out their testimonials and reviews in a wide array of directories and catalogs like Clutch and GoodFirms.
- Consider company websites and their experience. Are there trustworthy case studies in health technology, and does their website feature trustworthy customer reviews?
- Where is the company located? Are they HIPAA-compliant? Do they have a real office, or are they more like a fake IT agency hiring developers on gig platforms?
- Arrange a call with their representatives, including development leads. Make sure you’re represented by an experienced tech specialist (CTO) who can verify whether or not a potential vendor’s skills and capacities fit your project.
| ? If you’re interested in getting a free tech consultation or a plan estimate of medical device software development services, like IT team augmentation, please contact TATEEDA GLOBAL. We’d be happy to help! |
TATEEDA GLOBAL’s Experience in Creating Healthcare Equipment Software
We have completed a number of relevant projects, including the integration of healthcare devices with custom software components, embedded programming, API integration, client-server architecture, and more.
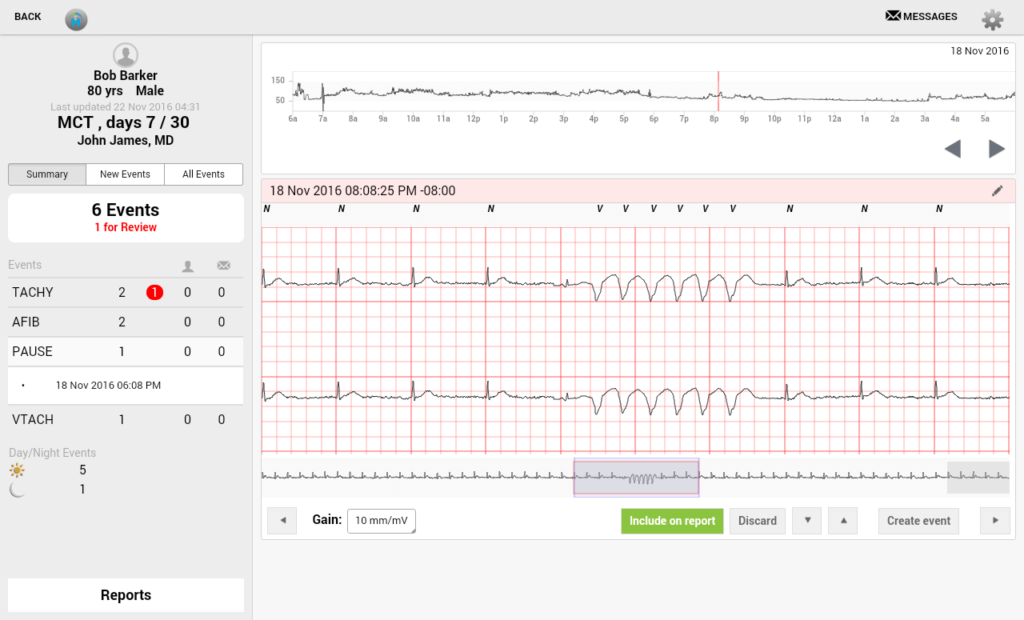
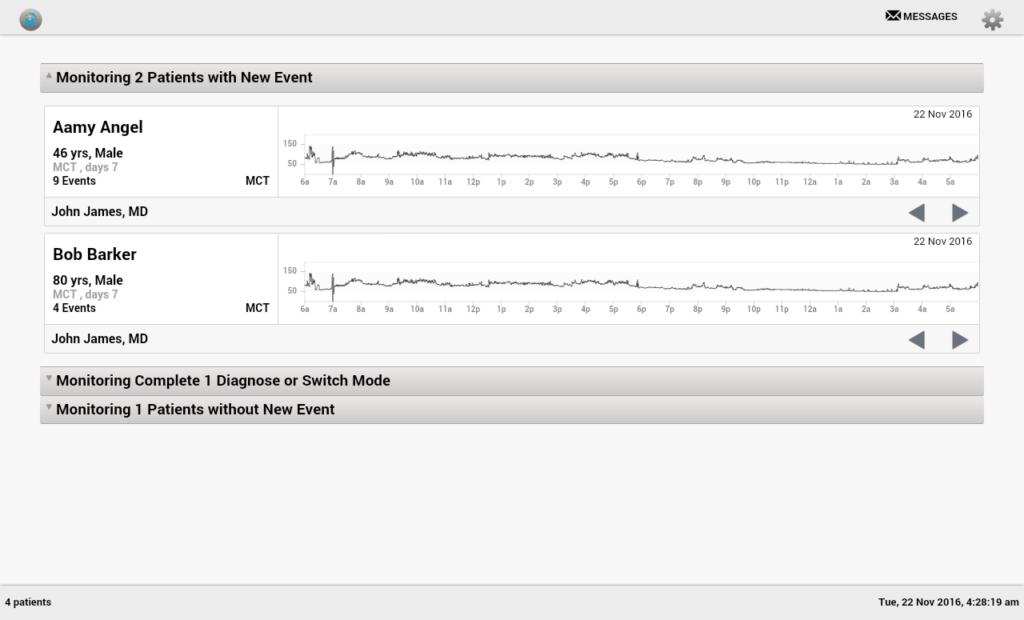
Example #1: Medical Device Software Design for Remote Cardiac Monitoring
One of our medical device app development projects was done for Ventrilink. The medical device in question was an ECG-monitoring IoT biosensor for remote patients. We helped Vetrilink…
- Establish a stable connection/interaction between the device and software components
- Optimized the server for optimum information intake
- Built a fast tablet application with real-time scalable ECG visualizations and many other vital functions. Learn more here.
Software design for medical devices includes multi-functional, convenient user interfaces, which embrace a sufficient set of pro-grade functionalities for physicians and other medical professionals:
Example #2: PC-based Software Development for a Medical Laboratory Device
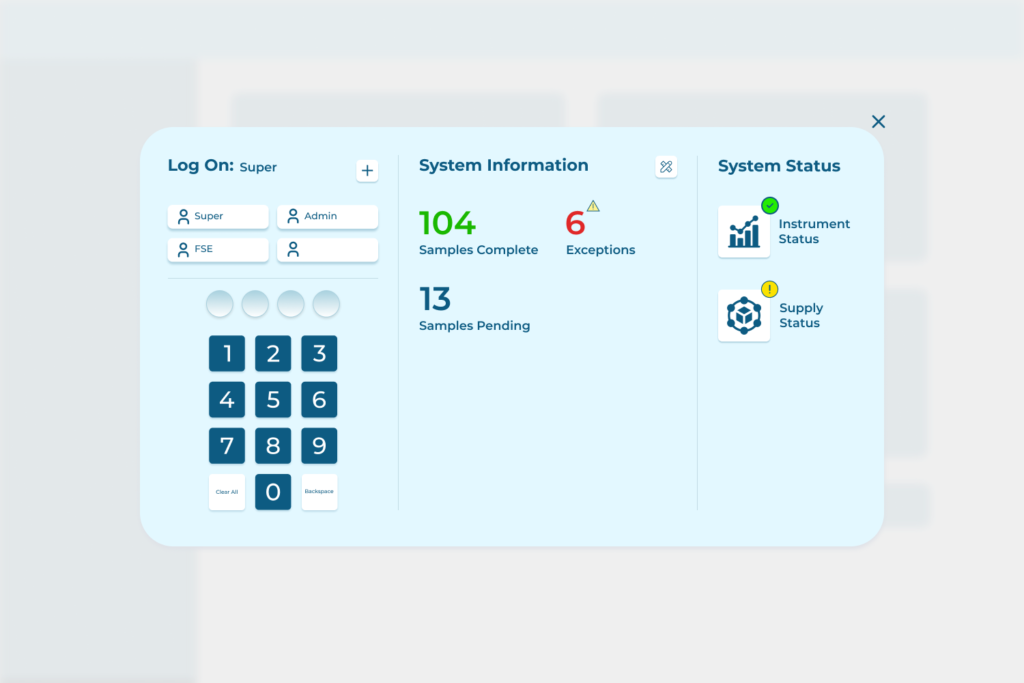
Another of our projects was executed for one of the world’s top manufacturers of lab equipment (cannot be named due to our NDA).
Our medical device software development services provided to this client include:
- Created a highly reliable desktop application that featured integration with low-level device components and high-level lab automation features
- Helped to build a status management system for samples being tested in the device, which enabled operators to control and visualize the queue of test tubes and containers.
- Designed interfaces to display important test parameters and results: electronic tables, device management menus, and scientific workflow indicators. Learn more here.

In Conclusion: Hire Software Developers for Medical Devices
If you need a professional IT team to partner with you on your custom medical device software development and/or project augmentation, TATEEDA GLOBAL is here for you!
We offer:
- Full-cycle development of healthcare-related software systems
- Equipment and IoT software project assistance, augmentation, and product testing
- Legacy device software upgrades and reengineering
- Long-term medical software maintenance services
- Affordable project costs, thanks to our R&D branch in Ukraine
- Personalized connection with our project manager HQ in San Diego, CA
- Vast onsite experience with U.S.-based IoT, healthcare, biotech, and pharma companies
- Fast team deployment—within 48-72 hours
Contact TATEEDA GLOBAL today and outsource your healthcare device software development to us!











